
Non-Sulfide Zinc Deposits:
a new-(old) type of economic mineralization
Maria Boni

It is not surprising that the Issue 98/4 (2003) of Economic Geology will be dedicated to non-sulfide zinc deposits, and that one session of the 7th Biennial SGA Meeting at Athens in August 2003 will also revolve on this subject. Indeed during the last years this type of ore deposits has been increasingly arousing the interest of mining companies and of the scientific world alike.
Prior to the development of flotation and smelting processes for zinc sulfide ores at the beginning of the 20th century, the non-sulfide deposits (often known collectively as "Zinc Oxides") were the principal source of zinc in the world. From Roman times up to the 18th century, the non-sulfide Zn-ores, a mixture of silicates and carbonates known as "Lapis Calaminarius", "Calamine", "Galmei", or "Galman", in the Latin-, French-, German-, and Polish-speaking world respectively, were used as the source minerals for the production of brass, a zinc-copper ±tin alloy fairly widespread throughout Europe and the Mediterranean area over the centuries (Boni & Large, 2003). đ Later, the non-sulfide ores were processed to produce high-grade zinc oxide in Wälz kilns, using a technology that was discovered in Belgium and then developed throughout Europe during the 19th century. Jean-Jacques-Daniel Dony, a chemist from Liège, invented the first process, patented in 1810 by Napoléon I, to transform the ores from the rich Belgian deposits (notably from "La Calamine", Moresnet) into malleable zinc. The metallurgical transformation of the "calamine" concentrates was done in horizontal crucible melting furnaces, operating for the first time in a factory at the Saint Léonard wharf in Liège (DeJonghe, 1998).
Table 1: Mineralogy commonly associated with non-sulfide Zinc deposits (modified from Large, 2001)
| Mineral | Composition (approximate) | Common Associations | Comments |
| Smithsonite | ZnCO3 | Found in most deposits, both supergene and hypogene | Common component of "calamine" |
| Hydrozincite | Zn5(OH)6(CO3)2 | Present in many deposits; recent, might replace smithsonite | Known as zinc "bloom" |
| Franklinite | ZnFe2O4 or (Fe,Zn,Mn)(Fe,Mn2)O4 |
Rare - principal mineral at Franklin/Sterling Hill | Zn-spinel |
| Gahnite | ZnAl2O4 | Common in Proterozoic metamorphic terranes often associated with massive sulfides | Zn-spinel |
| Hemimorphite | Zn4Si2O7(OH)2.2H2O | Present in many deposits - common in the upper part of the calamine orebodies | Common component of "calamine" |
| Sauconite | Na 0.3 (Zn,Mg)3 (Si,Al)4.OH2.nH2O |
Present in many deposits - typical of deposits associated with silicoclastites (es. Skorpion) | Zn-saponite - slightly different formulae reported by different AA. |
| Willemite | Zn2SiO4 | Typical of hypogene deposits, but occurring also in supergene ones (es. La Calamine, Belgium) | As hexagonal prisms or concre-tional, cryptocrystalline structures |
| Zincite | ZnO | Occasional, but principal mineral at Franklin and Sterling Hill, USA | Rare |
| Cerussite | PbCO3 | Occasional in the upper oxidation zones of many carbonate-hosted deposits | Occurring at Tynagh; dominant at Broken Hill |
| Anglesite | ZnSO4 | Occasional in the oxidation zones of many complex sulfide deposits | Only in supergene deposits |
| Pyromorphite | Pb5(PO4,AsO4)3Cl | Typical of supergene deposits where the alteration is on As-minerals | Can be confused with Mimetite |
| Mimetite | Pb5(AsO4,PO4)3Cl | Typical of supergene deposits where the alteration is on As-minerals, but occurring also in hypogene ones | Occurring at Angouran |
| Malachite Azurrite |
Cu2(OH)2(CO3) Cu3(OH)2(CO3)2 |
Occasional in the oxidation zones of deposits containing Cu-sulfides | Occurring at Tynagh, Skorpion and Berg Aukas (both hypogene and supergene) |
| Goethite Hematite |
FeO(OH)Fe2O3 | Ubiquitous in the oxididation zone of supergene deposits | Very common; can be used for paleomagnetic studies |
| Coronadite Hetaerolite Hollandite |
(Pb,Ba,K)1-2 Mn8O16.xH2O ZnMn2O4 (Ba,K)1-2 Mn8O16 .xH2O |
Mn-minerals generally occurring in hypogene, but occasionally also in supergene deposits | If containing K, suitable for absolute dating with K-Ar and 40Ar-39Ar methods |
| Ferroan dolomite | Ca(Mg,Fe)(CO3)2 | Typical of hypogene fracture-controlled willemite deposits | Occurring at Vazante as a praecursor hydrothermal phase |
With the development of solvent-extraction (SX) and electro-winning (EW) processes, and with the modernization of the Wälz technology for the treatment of non-sulfide zinc ores, there has been a renewed commercial interest for this style of mineralization throughout the world (Large, 2001; Boni & Large, 2003; Borg et al., 2003; Hitzman et al., 2003). The commercial exploitation of "Zinc Oxides" deposits is rapidly becoming an important source of metallic zinc and within the foreseeable future the annual production of zinc from oxide ores could vastly exceed 10% of the global zinc metal production (Table 1 in Large, 2001; Tables 1 and 3 in Hitzman et al., 2003). Today, the attraction of these deposits includes the scale economy (individual projects mostly exceed 100.000 tons zinc metal) and the projected low processing costs for the production of zinc metal or high-grade zinc oxide on site (Large, 2001). Compared to sulfide deposits, their main attraction lies in: a) their distinct scarcity or lack of Pb, S and other undesirable elements, b) their relatively low-energy recovery by SX-EW, and c) the generation of higher economic value on site.
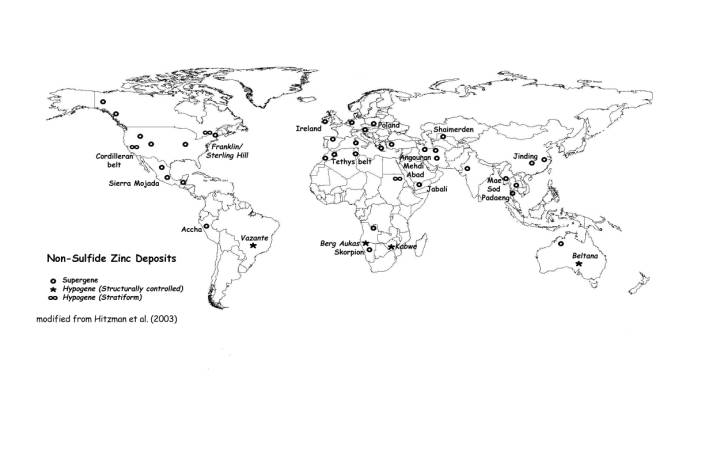
Tonnages in the deposits range from < 1 Mt to > 200 Mt with grades of 7% to more than 30% Zn (Large, 2001; Hitzman et al., 2003; Reynolds et al., 2003). Deposits of this size include Skorpion (Namibia), Mae Sod (Thailand), Lan Ping (China), Angouran (Iran), Mehdi Abad (Iran), Shaimerden (Kazakhstan), Jabali (Yemen), Sierra Mojada (Mexico) and Franklin/Sterling Hill (USA). In addition, there are a number of other mines producing relatively small tonnages of non-sulfide zinc ores in Vietnam (Cho Dien), Turkey, China, Morocco and Egypt. For the sake of completeness, one should add to the total the high tonnages recovered in formerly exploited districts, like SW Sardinia, Belgium and Ireland, as well as in the areas where the non-sulfide zinc concentrations are not considered a resource, like in Upper Silesia (Poland) (Boni & Large, 2003). The location of some of the most important deposits throughout the world, is shown on Figure 1.
Scientific research, resulting from the economic interest in this class of mineralization shown recently by many mining companies, has been revived after more than fifty years of neglect of what most scientists, including this writer, have for long considered a non-economic "rubbish" which, in the most favorable case, could have been only an important indication in the exploration of a hidden sulfide primary ore.
Research is now being focused not only on several economic "Zinc-Oxide" deposits throughout the world (e.g., Skorpion, Namibia: Borg et al., 2003; Shaimerden, Kazakhstan: Boland et al., 2003; Vazante, Brasil: Monteiro et al., 1999, Hitzman et al., 2003; Beltana, Australia: Muller, 1972, Groves & Carman, 2003, Hitzman et al., 2003; Angouran, Iran: Hirayama, 1986, Gilg et al., 2003), but also on older mining districts containing smaller, historically exploited deposits (Boni et al., 2003; Boni & Large, 2003; Johnson & Skinner, 2003).
The aim of these studies is not only to understand the geological constraints on the distribution of this kind of orebodies together with their timing of formation, but to reach also a better definition of the mineralogy and geochemistry of the mineral phases (Aversa et al., 2002; Boni et al., 2003; Brugger et al., 2003). A thorough understanding of the relationships of mineralogy (Table 1) to deposit type, may significantly aid in exploration targeting. In fact, although the hydrometallurgical behavior of the non-sulfide zinc minerals is only poorly described in the modern scientific literature (there are more than abundant papers on this subject until the seventies), recent unpublished studies indicate that most non-sulfide zinc minerals, with only few exceptions, are easily leachable in sulfuric acid, allowing recoveries of >94% Zn (Hitzman et al. 2003).
Although there are many similar features described in the past for most non-sulfide zinc deposits, the only existing classification of this kind of ores until recently was due to Heyl and Bozion (1962), and was mostly limited to the deposits in the United States.
Large (2001) describes three distinct classes of deposits, based on the style and setting of the mineralisation, as well as on the dominant mineralogy (Fig. 2).
These are:
1. "Calamine"-dominated deposits in Mississippi Valley Type and other stratiform sulfide primary ores in carbonate rocks. Here the non-sulfide mineralisation is related to oxidation of primary sulfides and preservation in karst-cavity in-filling and replacement aggregations;
2. Willemite-dominated deposits in late Proterozoic to early Cambrian sedimentary rocks, where the mineralisation occurs in marked fault zones. These deposits might be hydrothermal in origin, formed under specific low S- and high O-fugacities;
3. "Gossan"-type deposits, containing hydrated zinc silicates that were formed by residual surface oxidation of primary sulfides and then preserved by a special set of circumstances (tectonic, climatic, etc.).
Hitzman et al. (2003) have produced a more articulated classification, in which a broad distinction between supergene and hypogene deposits has been contemplated for the first time. The supergene deposits, corresponding to types 1. and 3. of Large (2001), consist mostly of the economic minerals smithsonite, hydrozincite and hemimorphite. They form primarily from the oxidation of sulfide-bearing deposits (Fig. 2) and can be subdivided into three main subtypes:
Many, but not all, of the oxide deposits of supergene type, are located between latitudes 15° and 40°N (Fig. 1), and this may reflect particularly favorable climatic conditions conducive to formation of secondary zinc minerals. However, it is possible that most of the existing deposits (e.g., the European "Calamine", Boland et al., 1992; DeJonghe, 1998; Boni & Large, 2003; Boni et al., 2003) have been rather due to paleoweathering episodes post-dating important emersion phases.
The critical geological features for the oxidation of primary sulfides and preservation of the secondary zinc minerals might include:
There is insufficient space to review the geology of numerous individual supergene non-sulfide zinc deposits, and the reader may refer to the literature cited (check mostly Hitzman et al., 2003). However, one can enumerate here the characteristics of the most important mineral occurrences among those belonging to this category.
The Shaimerden supergene deposit in Kazakhstan (Boland et al., 2003), consisting of an irregular 300 x 200 m body extending to a depth of over 100 m, was formed by a combination of both wallrock and direct replacement. The deposit is hosted in Lower Carboniferous carbonates occurring in a volcanic-dominated succession, and is overlain by about 40 m of Cretaceous to Quaternary cover. The ore minerals consist mainly of hemimorphite, smithsonite, and minor sauconite.
The Cho Dien district in Vietnam is a good example of supergene mineralization formed by residual karstic processes (Hitzman et al., 2003). A number of small high-grade sulfide bodies related to a Triassic granite intrusion in Devonian metasediments, was mined in the past on a high karstic plateau rising 700 m above a valley floor. Residual supergene mineralization (grading up to 10 to 30% zinc) occurs in the cavities of the same plateau and, to a lesser extent, in transported colluvial overburden. Ore minerals consist dominantly of hemimorphite, minor hydrozincite and smithsonite.
In SW Sardinia (Italy) several styles of "calamine" mineralization have been recognized throughout the district (Fig. 3a), including partial replacement of the host carbonates and stratabound primary sulfides, as well as concentrations of ferruginous, "earthy" smithsonite and hemimorphite-rich clays (Boni et al., 2003). The mineralogy of the ore is dominated by smithsonite (Fig. 3b), hemimorphite (Fig. 3c) and hydrozincite, with in-situ grades generally higher than 20% Zn. The inter-relationships between weathering, uplift and erosion in the formation and preservation of non-sulfide zinc mineralization in SW Sardinia, have been analyzed by Moore (1972) and Boni et al. (2003). Both concluded that the oxidation of primary sulfides, commonly extending to a depth of several hundred meters below the surface, show no apparent relationship to the present water table and is probably related to Tertiary and even Mesozoic geomorphologic conditions (paleoweathering).
The Skorpion deposit in Namibia is a rare example of a wallrock-replacement deposit (Borg et al., 2003) derived from a pyritic volcanogenic massive sulfide orebody, hosted in a volcano-sedimentary succession of Neoproterozoic age. Uplift and erosion of the host rocks and of the massive sulfides were post-Damara orogen, thus allowing the circulation of oxidizing fluids, which then deposited zinc-silicates and -carbonates in the secondary porosity of the same volcano-sedimentary sequence. The timing of the formation of the deposit was possibly quite extended, synchronous with the discrete phases of Tertiary erosion on the pan-African continent. The mineralogy of the supergene zinc mineralization at Skorpion is dominated by hemimorphite and sauconite with lesser smithsonite. The supergene body appears to have formed primarily at the contact between carbonates with volcanic and clastic rocks. The non-sulfide ore minerals are undeformed, and have been precipitated in open spaces and small veins (Borg et al., 2003). Part of the secondary permeability was caused by the breakdown of feldspars and mica. In addition, the high feldspar content of the volcanoclastic units has contributed to the high proportion of zinciferous clays in the Skorpion deposit. The geometry and mineralogy of the deposit suggest that its main genetic process is largely due to wallrock replacement. However, the occurrence of partially oxidized sulfides in the felsic ore horizon down to depths of 800m shows that also in situ oxidation was present, and that the deposit can be partially attributed to the direct-replacement type.
The Angouran deposit is one of the largest of a series of carbonate-hosted mixed oxide/sulfide zinc deposits that occur within the 1600 km long Tertiary-age Zagros collisional belt of Iran (Hirayama, 1986; Gilg et al., 2003). It is hosted by a Neoproterozoic metamorphic complex consisting mostly of marbles and schists, overlain in turn by a Tertiary-Quaternary sedimentary and volcanic sequence. Estimated resources are more than 15 Mt ore at ~26% Zn and ~4% Pb. Angouran is not simply a supergene deposit, its characteristics pointing to a mixed supergene-hydrothermal origin, the latter related to fluid circulation during volcanic activity. The deposit consists of an oxide cap, underlain by a mixed sulfide-oxide body which grades downward into a sulfide body. The upper levels of the orebody are dominated by smithsonite-rich "oxide" ores (Gilg et al., 2003). Textures are variously soft and earthy, botroidal to crustiform, multiply brecciated, travertine-like, and cavernous with abundant open spaces and voids. The carbonate-oxide ores are products of supergene weathering. Mn-poor smithsonite, Fe-Mn-oxides/hydroxides, Zn-rich clays, mimetite, calcite, hydrozincite and hemimorphite have been recorded in the weathering phases. In proximity to the footwall schist, mixed "oxide"-sulfide ores (Fig. 3d), and then sulfide-dominated ores are found. The "oxide"-sulfide ores are composed of smithsonite and accessory, co-precipitated arsenopyrite, galena, pyrite and quartz. Gilg et al. (2003) suggest that the carbonate-sulfide mineralization at Angouran was deposited by a distinct low-temperature hydrothermal system, most probably related to Tertiary-Quaternary volcanic activity.
The hypogene deposits, which have been only recently recognized as a new type of zinc ore (Hitzman et al., 2003; Large 2001, type 2.), consist dominantly of zinc silicates and oxides with minor sulfides (Fig. 1). Willemite (Table 1) with pseudo-colloidal textures instead of smithsonite or hemimorphite, seems to be the most characteristic mineral in this class of deposits. Ore deposition has been considered to occur by mixing a reduced, high temperature (>80° to <200°C) zinc-rich, sulfur poor fluid, with an oxidized sulfur-poor fluid. The latter could have been seawater, groundwater, or a basinal fluid, which has equilibrated with an oxidized rock mass, such as a red bed sequence or a weathered regolith (Hitzman et al., 2003). The hypogene deposits have been subdivided into two subtypes:
To the first subtype belong, among others, the Vazante-Morro Agudo (Brasil), Beltana-Aroona (Australia), Kabwe (Zambia) and Berg Aukas (Namibia) deposits, which all share a number of common characteristics. They are structurally-controlled, forming veins or pipe-like bodies along normal faults and have variably developed halos of hydrothermal dolomitization, preceeding a major period of willemite precipitation. They also have a mineral assemblage which ranges from solely willemite at Beltana (Groves & Carman, 2003), to willemite-(sphalerite) at Vazante (Monteiro et al., 1999) and Berg Aukas (Chadwick, 1993) and to sphalerite-(willemite) at Kabwe (Kamona, 1993), suggesting that these deposits might represent a continuum (Hitzman et al., 2003). The most outstanding example of this class of deposits, the Vazante mine, is located in the Neo-Proterozoic succession of the state of Minas Gerais in Brasil (Fig. 1). The main resources consist of unusual willemitic ore stringers located along important structural lineations, with small sulfide bodies imbricated with the willemite (Fig. 4). Monteiro et al. (1999) show that the relationships between sphalerite and willemite at Vazante are complicated by the deformation on the Vazante shear zone, but suggest also that the willemite mineralisation should have occurred under conditions of unusual high O- and low S-fugacity during deformation (Large, 2001). In fact, the structural relationships and mineral chemistry at Vazante suggest that the mineralisation may have occurred during, or shortly after, the first deformation of the host sedimentary succession from fluids expelled from the sedimentary succession to the site of mineralisation along major cross-stratal structures. In addition to the "primary" willemite mineralisation, in the same deposit variable amounts of near-surface calamine also occur, as late replacement and cavity-fillings in host carbonates.
The hypogene stratiform non-sulfide zinc deposits appear to be a rare type of deposit (Fig. 1) and the origin of the Franklin and Sterling Hill metamorphic orebodies, which are the most characteristic of this sub-class, is still controversial. Both the above quoted mines, occurring in New Jersey (USA), are now closed but were a very important source of zinc in the past. The Franklin District, where the mineralisation was hosted by Grenvillian Upper Proterozoic marbles, produced 23 million short tons of ore (ca 20.86 Mt) from 1850 to 1954at an average grade of 19.5% zinc. Ore minerals consist of franklinite, willemite and zincite, in layers concordant with the metamorphic foliation. In the older literature, many authors proposed that prior to metamorphism, the deposit consisted already of secondary minerals, such as hemimorphite and hydrous Mn- and Fe-oxides, derived from the oxidation of preexisting sulfides. Johnson (2001), however, cautiously advances the concept that the Franklin and Sterling Hill deposits are extremely rare occurrences of exhalative zinc carbonate-silicate oxide accumulations in a sulfur-depleted basin, and in this they may have some similarities to the so-called Broken Hill-type deposits.
In Johnson & Skinner (2003), there is a paramount view of the genetic theories on this kind of deposits, as well as an interpretation of the famous "magnetite beds" of Franklin Furnace.
However, although the newly defined hypogene deposits might be interesting, the economic focus seems to lie rather on the supergene ones, with the highest promising areas located in the Asiatic continent. A major problem for this class of deposits is that the time constraints for the deposition of many, apparently supergene, non-sulfide ores are still unclear, due to multiple oxidation events through time, controlling in turn the variable paragenesis of Zn-silicates/carbonates. Even if not considering the geologically complex deposits, like Angouran, where part of the non-sulfide assemblage seems to be rather related to primary hydrothermal segregation instead of being a weathering product (Gilg et al., 2003), there is enough evidence that also many other 100% supergene orebodies, might be recording several episodes of paleoweathering through time. Because the timing and evolution of the weathering profiles in areas with hypothesized sulfide primary ores, might be having interesting implications for the exploration (and evaluation) of this type of ore in many parts of the world, it remains a priority to try to obtain a direct age of the oxidation phenomena, either by paleomagnetic methods or by using radiogenic isotope systems, such as 40Ar-39Ar or U-Th-He on K-Mn-oxides.